In the Laboratory
- A brief history of tissue processing
- Errors in the histology laboratory
- Lean and learn - efficiency in the histology laboratory
- Objectives of cellular pathology
1
A brief history of tissue processing
A brief history of tissue processing
Since the word ‘tissue’ was defined in the late 1700s by Marie Francois Bichat, the father of histology, the advances that have been made in the world of cellular pathology are beyond measure. Though his original work was based on the gross dissection of tissue samples, modern histology with its microwave processing, automation for cutting and staining, microscopy and digitalization of whole slide images is a world away from the primitive laboratories of Bichat. Nonetheless, while these new techniques and discoveries have improved the way that histology has evolved, certain aspects of the science have essentially remained unchanged. Formaldehyde for example was discovered in the mid 1800’s yet it still remains the gold standard for tissue fixation.
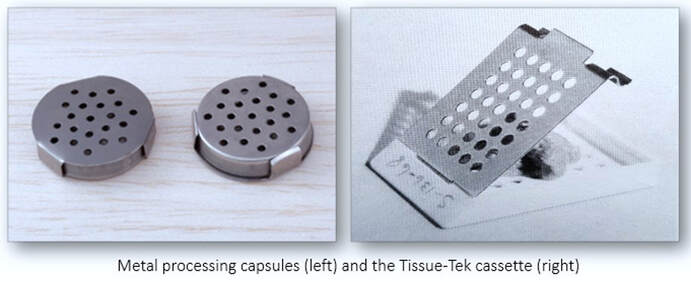
One of the first tasks of any histology laboratory is the receipt, recording and labelling of tissue samples on their arrival. Often termed accessioning, the accurate numbering of specimens is an essential component for the identification of patient samples. Long before the age of the computer, laboratories relied on the hand numbering of samples and request forms prior to recording their entry into the laboratory ledger. Numbered labels were often written in pencil onto small oblong pieces of paper and placed with the specimen in formalin so that they remained with the correct sample throughout. Once specimens had been successfully numbered and logged, careful examination and selection of tissue samples for processing were carried out in the dissecting or grossing room. Historically, the tissue samples together with their hand-written identification labels were placed into perforated, stainless steel capsules. Many years later, advancements were made with the introduction of the Tissue-Tek processing cassette. This system not only enabled the tissue samples to be safely secured in permanent plastic cassettes but also allowed the specimen number to be hand written on one edge.
Since paraffin wax was first introduced as an embedding medium by Edwin Klebs in 1869, it has been used extensively as an infiltration and support medium for the sectioning of tissue blocks. Nonetheless, because of the hydrophobic nature of the wax, the requirement for tissues fixed in aqueous formalin to be dehydrated in alcohol and cleared through changes of solvent such as xylene still remains. In the early days, tissue processing would have been carried out by hand but this was superseded by the introduction of the automated tissue processor around the middle of the 20th century. With tissue transfer processors, the capsules or cassettes containing the tissue samples were placed into perforated baskets and the tissues transferred from reagent to reagent. This was carried out through a series of stationary reagent containers arranged in-line or in a circular, carousel arrangement. Processing schedules were generally cut into notched discs and fluid agitation was achieved either by a rotary motion of the basket or by vertical oscillation. Although the original models were open, stand-alone types, more recent models had integrated fume hoods. Towards the end of the 20th century, fluid transfer processors had evolved in which the processing fluids were pumped to and from a sealed retort in which the tissues remained stationary. These systems were totally enclosed and filters were in place to absorb any vapours that arose from the processing reagents.
Once tissues had finally been infiltrated with molten paraffin wax, they were transferred to moulds for casting or blocking. This stage is more commonly known as embedding and over the centuries, various types of mould have been employed. In the distant past when hand-written labels were commonplace, glass or metal Petri dishes smeared with glycerine were utilized for embedding several pieces of tissue at a time. While paper boats were both suitable and cheap, glass test tubes and watch glasses were sometimes utilized although they often required breaking in order to release the tissue block. Although plastic ice cube trays were a useful addition, adjustable moulds in the form of Leuckhart ‘L’ pieces were popular at the time because they could be adapted to form an oblong mould which would suit any size of tissue sample. These moulds consisted of two ‘L’ shaped metal pieces (usually brass) which were placed onto a metal or glass plate. Nonetheless, whatever the type of mould used, molten wax would be poured into it and the tissue transferred from its capsule or cassette. Any accompanying identification label would be placed into position at the edge of the mould as the wax was solidifying. Once cooled, the tissue block would be removed and the excess wax trimmed off. The block and label would then be mounted onto a wooden chuck in readiness for microtomy using a hot iron or spatula to melt the wax.
How times have changed! Since the introduction of stainless steel moulds and tissue cassettes, the need for transferring hand-written labels during processing and embedding has been laid to rest. During the last fifty years, many changes have been made to consumables alone in the histology laboratory. From plastic embedding rings to colour-coded standard, mega and mothership cassettes, production of the paraffin block still remains the primary foundation for histological study. Even with all the technological advances that are present in the modern histology laboratory, the paraffin block remains crucial to its future success.
Further reading
1. A historical review of paraffin and its development as an embedding medium (Sanderson C et al). J Histotechnol 1988;11:61-63
http://www.tandfonline.com/doi/abs/10.1179/his.1988.11.1.61?journalCode=yhis2
2. A short history of histopathology technique (Titford M). J Histotechnol 2006;29:99-110
http://www.tandfonline.com/doi/abs/10.1179/his.2006.29.2.99
3. Once upon a microscopic slide: The story of histology (Hussein IH et al). J Cytol Histol 2015;6:377
https://hsrc.himmelfarb.gwu.edu/smhs_anatregbio_facpubs/96/
4. Past, present and future: Overview on histology and histopathology (Musumeci G). J Histol Histopathol 2014;1-5
http://www.hoajonline.com/histology/2055-091X/1/5
Further reading
1. A historical review of paraffin and its development as an embedding medium (Sanderson C et al). J Histotechnol 1988;11:61-63
http://www.tandfonline.com/doi/abs/10.1179/his.1988.11.1.61?journalCode=yhis2
2. A short history of histopathology technique (Titford M). J Histotechnol 2006;29:99-110
http://www.tandfonline.com/doi/abs/10.1179/his.2006.29.2.99
3. Once upon a microscopic slide: The story of histology (Hussein IH et al). J Cytol Histol 2015;6:377
https://hsrc.himmelfarb.gwu.edu/smhs_anatregbio_facpubs/96/
4. Past, present and future: Overview on histology and histopathology (Musumeci G). J Histol Histopathol 2014;1-5
http://www.hoajonline.com/histology/2055-091X/1/5
2
Errors in the histology laboratory
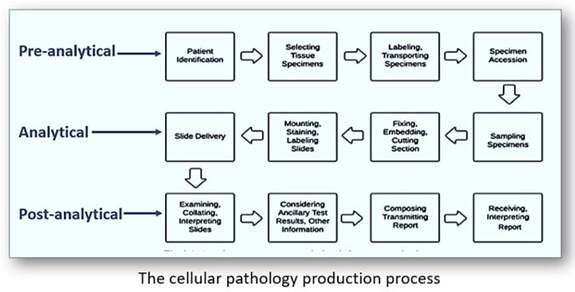
The primary role of the histology laboratory is the provision of a diagnostic service through the preparation, analysis and interpretation of tissue samples. Given that the procedures involved are so complex, it is inevitable that errors will transpire throughout the entire process. Identifying those errors and their root causes in the clinical setting can be perplexing. Whilst often partitioned into clerical, processing, technical and documentation categories, errors in cellular pathology are more traditionally subdivided into pre-analytical, analytical and post-analytical phases. Although there is often confusion as to what stages constitute each phase, pre-analytical phases are generally those processes that are involved from the collection of the tissue samples through to their arrival in the dissecting room following accessioning and data entry. The analytical phase relates to the actual carrying out of laboratory testing from tissue dissection through to the stained slide. Lastly, the post-analytical phase includes those actions that follow the analytical phase such as clinical interpretation, the pathology report and the regulation of storage, retention and disposal of tissue samples.
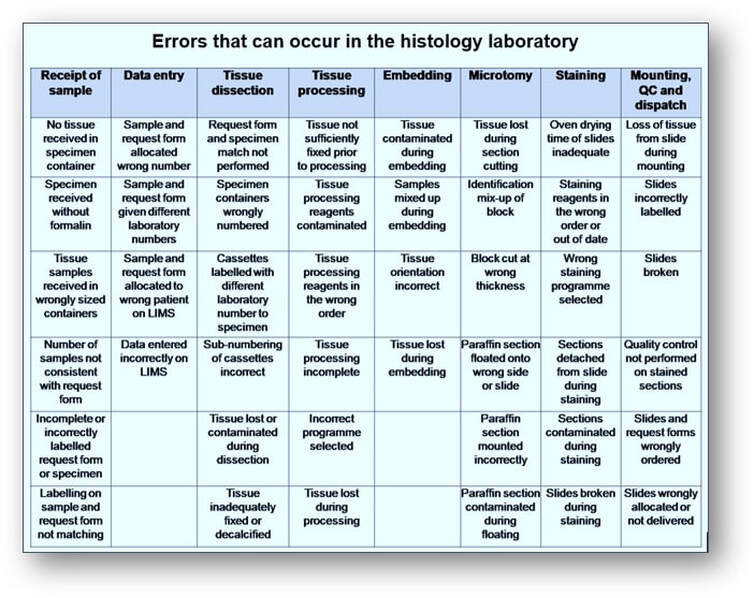
The pre-analytical phase encompasses many stages including the time prior to receipt of the sample in the laboratory (such as transportation from clinics and operating theatres). Since these stages regularly involve handling of the specimen by countless individuals, pre-analytical stages are related to one of the higher incidences of error and are often more complex to manage. Furthermore, the analytical errors that can occur in the histology laboratory during the preparation of a single haematoxylin and eosin stained and labelled slide from a tissue sample are so significant that the education and training of staff on their occurrences, causes and remedies is paramount (see Table below).
This process known as root cause analysis is a structured investigation often used as a means of identifying not only the source of errors but the actions that are required to eradicate them. Essentially, the purpose of it is to understand in detail what happened, why it happened and what could be done to prevent it happening again. To be effective, the methodology should be applied to all areas where errors can occur during the entire histological process from the receipt of sample to the typing and dispatch of the final histology report. Any information characterizing the occurrence of these errors is essential in providing strategies for both reducing and preventing them.
This process known as root cause analysis is a structured investigation often used as a means of identifying not only the source of errors but the actions that are required to eradicate them. Essentially, the purpose of it is to understand in detail what happened, why it happened and what could be done to prevent it happening again. To be effective, the methodology should be applied to all areas where errors can occur during the entire histological process from the receipt of sample to the typing and dispatch of the final histology report. Any information characterizing the occurrence of these errors is essential in providing strategies for both reducing and preventing them.
One of the areas that is particularly vulnerable to error in the histology laboratory is the manual entry of digital information whether it be onto laboratory information management systems, tissue cassettes or slides. Because histology sample numbers during accessioning are consecutive and composed of digits that differ only slightly from those found on adjacent samples, mistakes are inevitable. This predicament is often seen at the microtome where multiple blocks bearing similar numbers are found cooling on ice trays prior to sectioning, often onto pre-labelled, hand-written slides. This problem however, has largely been overcome with the introduction of bar-coded labels and scanners into the process, substantially decreasing errors in this area. Over the years, pathology laboratories have implemented a variety of control measures aimed at reducing the risk of cross-contamination of tissue. Many laboratories often avoid the serial numbering and dissection of similar specimen types; for example, gastric biopsies would be interspersed with other tissue types to reduce the risk of cross contamination with adjacent specimens. Also, the use of different coloured cassettes for specific tissue types can also help to minimize this problem. But without doubt, general tidiness and cleanliness of dissecting tools and benches, microtomes and water baths are often all that are required in preventing the cross contamination of tissue samples. Standard operating procedures pertaining to laboratory testing and analysis of tissue samples are in place as part of the training, accreditation and inspection practices. These standards together with appropriate internal and external quality control schemes are an integral part of quality management systems which together help to eliminate the root cause of errors in the histology laboratory.
The role of the pathologist
Although the histology diagnosis plays an important role in the treatment of disease, errors do occur. Diagnostic fallibility for instance, is an expression of error and when inaccuracies are present in the histology report, they can critically affect the outcome and care of the patient. Errors in fallibility are categorized either as oversights (where significant pathology is missed) or misinterpretations (where changes in pathology are interpreted incorrectly). While fallibility is an expression of error, credibility is the extent to which diagnostic opinions can be believed. In contrast, plausibility questions the consistency that exists between the histology diagnosis and the clinical picture. Excessive workload and human error are often common factors for potential in diagnostic fallibility. There is always the possibility that an error in diagnosis can give rise to damage accountability and the signature on the histology report will sanction that diagnosis. If the mistake was the consequence of an error of judgement (rather than the result of negligence or incompetence), it would show that the pathologist was exercising the same capability and understanding of that disease process as any other pathologist would have with the same level of knowledge and experience. Not all histology samples have the same diagnostic challenge and with many pathologists having different areas of speciality, it is often a difficult undertaking to be able to compare their levels of expertise. As part of a risk management strategy, referral of difficult cases, peer review auditing and attendance at multidisciplinary team meetings can all help to improve the diagnostic standards and hence minimize the risks of an incorrect diagnosis. Likewise, the monitoring of technical staff through training, continued competency and proficiency testing in the laboratory is essential in maintaining an error and risk-free environment.
Further reading
1. Achieving high reliability in histology. Improvement to reduce errors (Heher YK et al). Am J Clin Pathol 2016;146(5):554-560
https://www.ncbi.nlm.nih.gov/pubmed/28430956
2. Analysis of errors in histology by root cause analysis (Morelli P et al). J Prev Med Hyg 2013;54(2):90–96
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4718384/
3. Error and error reduction in pathology (Sirota RL). Arch Pathol Lab Med 2005;129(10):1228-1233
https://www.ncbi.nlm.nih.gov/pubmed/16196509
4. Errors: Detection and minimization in histopathology laboratories (Karki S). Journal of Pathology of Nepal 2015;5:859-864
http://www.nepjol.info/index.php/JPN/artic le/viewFile/15643/1260
5. Mislabeling of cases, specimens, blocks and slides. (Nakhleh RE et al). Arch Pathol Lab Med 2011;135(8):969-974
https://www.ncbi.nlm.nih.gov/pubmed/21809987
6. Specimen labelling errors in surgical pathology. (Layfield LJ & Anderson GM). Am J Clin Pathol 2010;134(3):466-470
https://www.ncbi.nlm.nih.gov/pubmed/20716804
The role of the pathologist
Although the histology diagnosis plays an important role in the treatment of disease, errors do occur. Diagnostic fallibility for instance, is an expression of error and when inaccuracies are present in the histology report, they can critically affect the outcome and care of the patient. Errors in fallibility are categorized either as oversights (where significant pathology is missed) or misinterpretations (where changes in pathology are interpreted incorrectly). While fallibility is an expression of error, credibility is the extent to which diagnostic opinions can be believed. In contrast, plausibility questions the consistency that exists between the histology diagnosis and the clinical picture. Excessive workload and human error are often common factors for potential in diagnostic fallibility. There is always the possibility that an error in diagnosis can give rise to damage accountability and the signature on the histology report will sanction that diagnosis. If the mistake was the consequence of an error of judgement (rather than the result of negligence or incompetence), it would show that the pathologist was exercising the same capability and understanding of that disease process as any other pathologist would have with the same level of knowledge and experience. Not all histology samples have the same diagnostic challenge and with many pathologists having different areas of speciality, it is often a difficult undertaking to be able to compare their levels of expertise. As part of a risk management strategy, referral of difficult cases, peer review auditing and attendance at multidisciplinary team meetings can all help to improve the diagnostic standards and hence minimize the risks of an incorrect diagnosis. Likewise, the monitoring of technical staff through training, continued competency and proficiency testing in the laboratory is essential in maintaining an error and risk-free environment.
Further reading
1. Achieving high reliability in histology. Improvement to reduce errors (Heher YK et al). Am J Clin Pathol 2016;146(5):554-560
https://www.ncbi.nlm.nih.gov/pubmed/28430956
2. Analysis of errors in histology by root cause analysis (Morelli P et al). J Prev Med Hyg 2013;54(2):90–96
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4718384/
3. Error and error reduction in pathology (Sirota RL). Arch Pathol Lab Med 2005;129(10):1228-1233
https://www.ncbi.nlm.nih.gov/pubmed/16196509
4. Errors: Detection and minimization in histopathology laboratories (Karki S). Journal of Pathology of Nepal 2015;5:859-864
http://www.nepjol.info/index.php/JPN/artic le/viewFile/15643/1260
5. Mislabeling of cases, specimens, blocks and slides. (Nakhleh RE et al). Arch Pathol Lab Med 2011;135(8):969-974
https://www.ncbi.nlm.nih.gov/pubmed/21809987
6. Specimen labelling errors in surgical pathology. (Layfield LJ & Anderson GM). Am J Clin Pathol 2010;134(3):466-470
https://www.ncbi.nlm.nih.gov/pubmed/20716804
3
Lean and learn - Efficiency in the histology laboratory
Lean and learn - Efficiency in the histology laboratory
Many years ago, a Japanese automotive engineer by the name of Taiichi Ohno helped create an efficient production system whilst working for Toyota. Based on the idea of increasing value for less work, any malfunctions in the system would be regarded as waste and all attempts to eliminate them would be made. This framework become known as Lean manufacturing and evolved from the Toyota production system with the help of other progressive development concepts such as Six Sigma, Kaizen and Kanban. Together, these manufacturing and production methodologies are globally accepted as the leading tools for efficiency and continuous improvement and can equally be applied to the clinical laboratory. Lean is a structured and methodical approach for solving problems by reducing the eight recognized categories of waste easily memorized by using the aptly named acronym DOWNTIME (see below).
In the clinical setting, provision of a reliable and efficient service for the analysis and diagnosis of tissue samples is the primary function of the cellular pathology laboratory. Modern technology is often combined with ageing practices and procedures so minor workflow improvements do not always benefit under those constraints. With sections of the laboratory often in different rooms or areas, layout is a major contributing factor to poor efficiency. Relocating equipment and reconfiguring these areas can help remove bottlenecks and provide a smooth transition between processes. Ending one process at the same place where the next one begins is the ideal scenario but cost, space constraints and safety of staff are often critical factors in reorganisation.
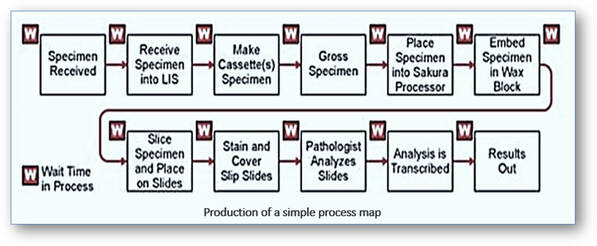
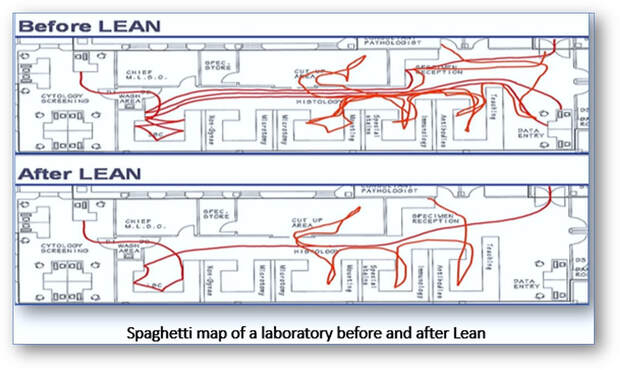
The application and maintenance of Lean principles help to improve workflow without compromising quality by recognizing areas of waste that impact on the overall efficiency of the histology laboratory. Probably the simplest way of identifying these areas is in the production of a workflow diagram or process map (see figure below). These are prepared by representing a visual journey of a sample by walking the flow through each process in the laboratory. Examining each step in turn and identifying areas of waste such as unnecessary waiting times and bottlenecks will help to assess the overall value and efficiency of the laboratory.
The application and maintenance of Lean principles help to improve workflow without compromising quality by recognizing areas of waste that impact on the overall efficiency of the histology laboratory. Probably the simplest way of identifying these areas is in the production of a workflow diagram or process map (see figure below). These are prepared by representing a visual journey of a sample by walking the flow through each process in the laboratory. Examining each step in turn and identifying areas of waste such as unnecessary waiting times and bottlenecks will help to assess the overall value and efficiency of the laboratory.
Progress of samples or staff can also be visualized by creating a continuous flow or spaghetti diagram on a plan of the laboratory before, during and after a Lean event. Recognizing redundancies and malfunctions such as excessive, repetitive or unnecessary movements in the system can be readily identified on spaghetti diagrams for particular laboratory processes or specific time spans.
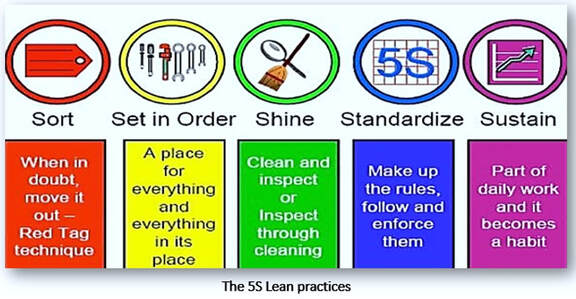
The organization and maintenance of orderly workspaces is also achievable using additional Lean practices known as 5S. Once completed, these areas help to reduce excess motion or movement by ergonomically designing and arranging equipment so that staff can interact with it both efficiently and safely. A sixth S for ‘Safety’ can also be designed into the mix by risk assessing any new processes prior to implementation. Also, inclusion of a flip chart on which staff can record recommendations and suggestions during an event is a useful adjunct in the laboratory during any Lean process.
So what are the benefits in designing a Lean laboratory? In the changing landscape of the Lean culture, transition of laboratory workloads to the pathologists and improvements in turnaround time has a direct impact on patient care. Elimination of processes that do not add value to the work and the introduction of many, sometimes small changes all contribute to that sustainable culture of continuing improvement. For decades, processing tissue samples overnight was the only alternative in the histology laboratory, but now same day results are achievable through the introduction of improvements such as faster biopsy processing and extended working days. Also, in laboratories where poor staffing is frequently an issue, Lean changes to workflow scheduling often help in releasing time for the development of task-specific competencies, training and regular staff meetings which under normal circumstances often prove difficult. Examination of other processes such as sample delivery times from the patient to the laboratory and the time periods taken from distribution of slides to the pathologists to issue of histology reports should also be reviewed. Improving patient care is at the forefront of diagnostic histology and only by maintaining a Lean culture that involves both technical and medical staff alike can efficiency be sustained without sacrificing quality.
Further reading
Further reading
- Experience and challenges in implementing Lean healthcare in pathology laboratory (Tajuddin A et al). Journal of Health Management 2022;(17(2):20-33. https://myjms.mohe.gov.my/index.php/johm/article/view/19154
- Guest editorial: Lean Six Sigma developments in Türkiye (Pakdil F et al). International Journal of Lean Six Sigma 2023;14(3):517-519. https://doi.org/10.1108/IJLSS-05-2023-231
- Reducing turnaround time in a pathology laboratory using the lean methodology (Akbulut Y et al). International Journal of Lean Six Sigma 2023;14(3):520-533. https://doi.org/10.1108/IJLSS-12-2021-0213
4
The objectives of cellular pathology
The objectives of cellular pathology
The principal role of the histopathologist is to provide an efficient and reliable diagnostic service. Through biopsy interpretation and assessment of surgical resections and autopsies, diagnostic testing can differentiate between normal and abnormal states. Making a diagnosis is a powerful cognitive process that involves knowledge and judgement. Knowledge is defined as the acquisition of information while judgement (along with intuition) is the skill which allows the pathologist to recognize and classify disease processes. These diagnostic skills are linked to the thought process (critical thinking) and in pathology can be outlined as follows:
- Knowledge of symptoms and disease processes.
- Understanding of the organs and tissues involved.
- Identification of the pathological processes present.
- Analysis and discrimination of one pathological process from another.
- Synthesis to determine the most probable cause.
- Evaluation by deciding the likely diagnosis.
The progress that has been made in biopsy methods have led to a more detailed understanding of many disease processes. With diagnosis being made on tiny fragments of tissue, a compromise must be found between the amount of tissue required for reliable interpretation and that which can be removed safely. Generally, selection of technique will often be determined by patient status, location of lesion, the equipment available and personal experience. The procurement and interpretation of an accurate biopsy specimen is one of the most important steps in the management of patients, often afforded by the participation of pathologists in clinical teams. This is achieved through multidisciplinary meetings where the clinical pictures and biopsy interpretations are presented and discussed with other hospital specialists. Biopsies can provide a diagnosis which helps to determine the extent of a particular lesion and predict biological behaviour. In ‘at risk’ patients for example, endoscopic surveillance can help monitor the likelihood of cancer development by detecting abnormal nuclear changes such as dysplasia, a change that is unequivocally neoplastic.
Disease, diagnosis, and competence
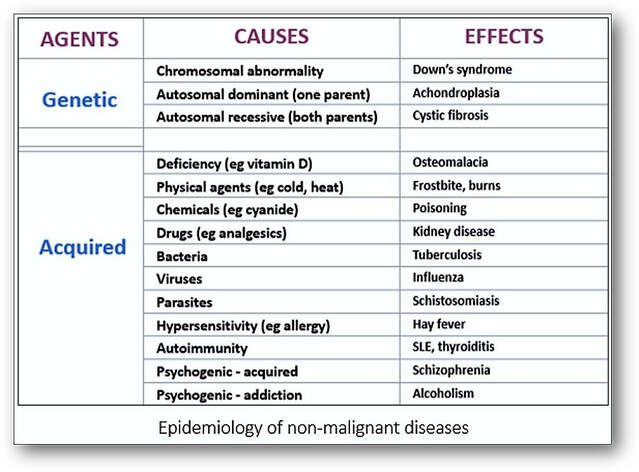
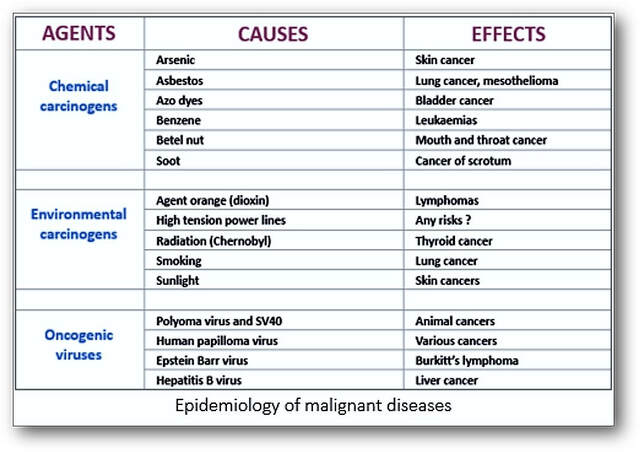
Studies of the incidence and prevalence of disease as it affects groups of people is termed epidemiology and originally dealt with epidemics of infectious diseases (such as smallpox) and outbreaks of infections (such as gastroenteritis). Nowadays, it is applied to widespread non-infectious diseases such as cancer and heart disease (see Tables below). Incidence of disease defines the extent or frequency of its occurrence, whilst prevalence indicates how widespread a particular disease is (prevalence = incidence x duration). Because of the large volume of tissue samples examined, histopathologists are ideally placed to study epidemiology. Classification of diseases with uniformly consistent nomenclature also makes it easier to collect and investigate patients with the same disease. The identification and determination of cancerous and precancerous lesions may also involve screening asymptomatic populations as seen with breast and cervical smear testing. Histopathologists require a broad-based knowledge and understanding of the pathological and clinical aspects of disease. They must be self-motivated, able to work alone and as part of a team, regularly make critical decisions, have problem-solving skills and good visual pattern recognition. Histopathologists regularly interact with other professionals during clinicopathological meetings where the diagnosis and clinical management of patients is discussed.
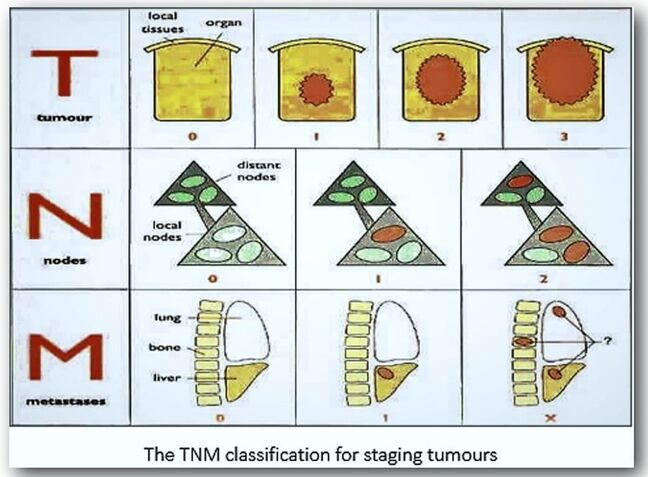
Making a diagnosis is the act of recognizing a disease from disorders in the structure and function of tissues, and it is these disturbances that are the hallmarks of a disease. Recognition of disease is accomplished by comparing the patient or lesion with what is regarded as the normal state. Although the art and skill of laboratory diagnosis generally lies within the realm of microscopy, tentative diagnoses can be achieved by the skilled pathologist and histologist from the macroscopic appearance of certain tissues in the dissecting room. Benign conditions such as lipomas, uterine fibroids and dermoid cysts can quite easily be recognised. Similarly, carcinomas of the breast, bowel, and ovary for example, can often be readily identified. A diagnosis is never made on macroscopic appearances alone but is always supported by microscopic examination, where the pathological process is assigned a name (papilloma, carcinoma) and tissue type (squamous, glandular). In tumour pathology, the degree of differentiation (grading) and depth of invasion (staging) are also major factors that help to determine treatment and prognosis. Grading is usually classified as low or high grade and describes the abnormality of the cells and the rate they are likely to grow. Staging on the other hand determines the size and spread of the tumour within the body. The most common staging system is the TNM classification where the ‘T’ score is based upon the tumour size and extent of invasion, the ‘N’ score indicates the extent of lymph node involvement, and the ‘M’ score indicates whether there are distant metastases present (see below).
Although histological diagnosis plays a vital role in the treatment of cancers and other diseases, errors can occur. These can take place at any level of the diagnostic pathway, and it is up to the clinician in charge of the case to interpret the full meaning of the biopsy result. Diagnostic fallibility is an expression of error, and it is these inaccuracies in histology reports that can critically affect patient care. Errors are categorized as oversights (where significant pathology is missed) or misinterpretations (where pathological changes are wrongly interpreted). These errors are further classified as Category 1, where the error would have a definite influence on clinical management and possible outcome; Category 2, where misinterpretation or oversight has the potential to affect clinical management or outcome, and Category 3, where a minor discrepancy of disease classification would likely to be of little clinical significance. While fallibility is the expression of error, credibility is the extent to which a diagnostic opinion can be believed. However, the term plausibility does not question how valid a particular diagnosis is but rather questions the consistency that exists between the diagnosis and the clinical picture.
Comparing individual pathologists is often a difficult task since most pathologists have specialist areas of interest. Individual histology samples may have differing degrees of challenge and uncertainty and as a result, audit will only have a limited role in assessing the competence of those histopathologists. Pathology departments should hold regular audits as part of a risk management strategy to help minimize the chances of an incorrect diagnosis. There are several options available to improve standards and these include clinicopathological meetings to maintain clinical liaison, peer review auditing using selected samples and specialist referral of difficult cases. The adoption of standard histological criteria and reporting guidelines contribute to quality standards by helping to reduce variations in interpretation of single samples by the same observer.
Diagnostic errors can give rise to damage liability, so it is essential that all procedures and circumstances surrounding a litigation event be documented sufficiently and that includes reception of samples, gross descriptions and microscopic interpretation. The signature on the final report endorses the diagnosis and all the statements in that report. The differences between what constitutes an error of judgement and that which is regarded as negligence or incompetence are clearly defined. For a mishap to be attributable to an error of judgement, it must be shown that the pathologist was exercising the same levels of competence and knowledge that would be shown by others of similar experience. Failure to apply those same general levels will expose the pathologist to the risk of successful litigation through negligence and incompetence. Human error and excessive workload are major contributory factors for the potential in altering patient management.
The role of the autopsy
For hundreds of years, dissection of the dead has been central to medical education. More recently, medical students were expected to participate in complete body dissections and to attend hospital autopsies as a means of teaching pathological concepts, clinicopathological correlations and anatomy. Even today, autopsies have a vital role in auditing medical care and perioperative deaths. Since many disorders are unrecognisable before death, comparing histological diagnoses with those from subsequent autopsies will help improve diagnostic methods and the degree of errors that occur in biopsy sampling. Discordance between primary clinical diagnosis and that obtained from autopsy has often found to be raised, particularly if tumours are malignant. Furthermore, in many patients, an undiagnosed tumour is often found to be an immediate cause of death. The decline of the autopsy in medical education has meant that fewer medical students are aware of its role in the teaching of medical fallibility and ethical practices in bereavement. Over many decades, the autopsy rate for patients dying in hospital has dropped steeply in many countries such as the United States, New Zealand and the United Kingdom. In addition, fewer medical schools require attendance at autopsy and those students graduate without attending a single session. One reason for the decline in the number of autopsies is an increased confidence in more modern methods of diagnosis, such as radiological imaging techniques and audiovisual teaching methods. Fear of malpractice, general apathy and controversies surrounding the retention of organs also play a role. Even when autopsies are performed, the information is often underused, with delays in reporting and lack of participation from clinicians. Autopsies should be the subject of external audit processes and clinicians should be involved in evaluating the quality of reports and the basis of conclusions and that includes causes of death.
Further reading
Comparing individual pathologists is often a difficult task since most pathologists have specialist areas of interest. Individual histology samples may have differing degrees of challenge and uncertainty and as a result, audit will only have a limited role in assessing the competence of those histopathologists. Pathology departments should hold regular audits as part of a risk management strategy to help minimize the chances of an incorrect diagnosis. There are several options available to improve standards and these include clinicopathological meetings to maintain clinical liaison, peer review auditing using selected samples and specialist referral of difficult cases. The adoption of standard histological criteria and reporting guidelines contribute to quality standards by helping to reduce variations in interpretation of single samples by the same observer.
Diagnostic errors can give rise to damage liability, so it is essential that all procedures and circumstances surrounding a litigation event be documented sufficiently and that includes reception of samples, gross descriptions and microscopic interpretation. The signature on the final report endorses the diagnosis and all the statements in that report. The differences between what constitutes an error of judgement and that which is regarded as negligence or incompetence are clearly defined. For a mishap to be attributable to an error of judgement, it must be shown that the pathologist was exercising the same levels of competence and knowledge that would be shown by others of similar experience. Failure to apply those same general levels will expose the pathologist to the risk of successful litigation through negligence and incompetence. Human error and excessive workload are major contributory factors for the potential in altering patient management.
The role of the autopsy
For hundreds of years, dissection of the dead has been central to medical education. More recently, medical students were expected to participate in complete body dissections and to attend hospital autopsies as a means of teaching pathological concepts, clinicopathological correlations and anatomy. Even today, autopsies have a vital role in auditing medical care and perioperative deaths. Since many disorders are unrecognisable before death, comparing histological diagnoses with those from subsequent autopsies will help improve diagnostic methods and the degree of errors that occur in biopsy sampling. Discordance between primary clinical diagnosis and that obtained from autopsy has often found to be raised, particularly if tumours are malignant. Furthermore, in many patients, an undiagnosed tumour is often found to be an immediate cause of death. The decline of the autopsy in medical education has meant that fewer medical students are aware of its role in the teaching of medical fallibility and ethical practices in bereavement. Over many decades, the autopsy rate for patients dying in hospital has dropped steeply in many countries such as the United States, New Zealand and the United Kingdom. In addition, fewer medical schools require attendance at autopsy and those students graduate without attending a single session. One reason for the decline in the number of autopsies is an increased confidence in more modern methods of diagnosis, such as radiological imaging techniques and audiovisual teaching methods. Fear of malpractice, general apathy and controversies surrounding the retention of organs also play a role. Even when autopsies are performed, the information is often underused, with delays in reporting and lack of participation from clinicians. Autopsies should be the subject of external audit processes and clinicians should be involved in evaluating the quality of reports and the basis of conclusions and that includes causes of death.
Further reading
- Visual thinking and the art of medical diagnosis (Chandler). In Design and Science (Editor L Atzmon), Bloomsbury Publishing 2023;14:287-300. https://doi.org/10.5040/9781350061958.ch-014
- Pearls and pitfalls in the diagnosis of small oral biopsies (McNamara & Kalmar). Semin Diagn Pathol 2023. https://doi.org/10.1053/j.semdp.2023.03.001